Ozone
Ozone has been successfully used in medicine because of its microbiologic properties for more than 100 years. Its bactericide, virulicide, and fungicide effects are based on its strong oxidation effects with the formation of free radicals as well as its direct destruction of almost all microorganisms. In addition, ozone has a therapeutic effect that facilitates wound healing and improves the supply of blood. For medical purposes, ozone may be applied as a gas or dissolved in water.
How much ozone is needed for disinfection? Ozone’s kill rate is proportional to its residual concentration. In one study, at 500 to 600 micrograms of ozone per m3 for one hour, 99% reduction in all bacterial specialities was observed.
The above studies were done by testing ozone against air-born bacteria. In fact, ozone kills bacteria 6,000 times faster than chlorine, when used in water.
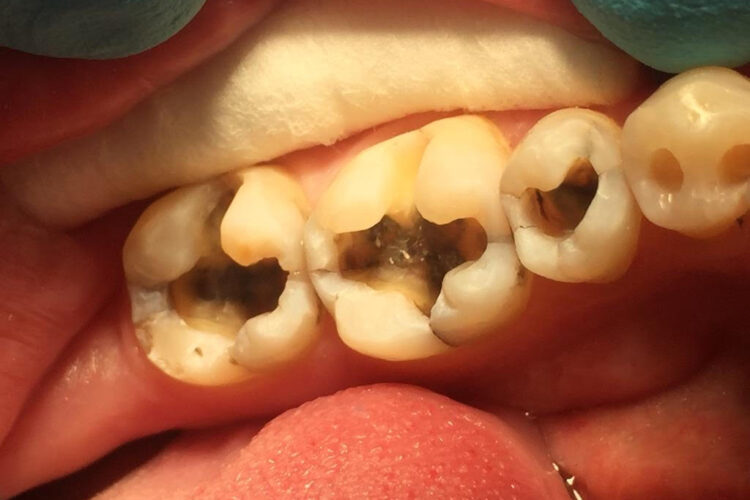
TEETH AFTER APPLYING OZONE
Dr. Ornelas was trained on O₃ (Ozone) by Dr. Renate Viebahn and Dr. Helmut Dorstewitz in 2005.

Dr. Renate Viebahn-Hansler
Scientific Consultant in Medical Ozone (Technology and Pharmacology)
Board Member of the International Ozone Association IOA; President Elect EA3G for 2023/24
Editorial board member of the scientific journal of the IOA: Ozone:Science & Engineering”.
Executive Board Member of the “Medical Society for Ozone Application in Prevention and Therapy”
Bacteria
When a high enough concentration of residual ozone (ozone that is not instantly reacting with substances in the environment) comes in contact with bacteria, the ozone reacts. Kinetic energy is released, and the single “activated” atom of oxygen is liberated, destroying the cell wall of the bacteria. Once the cell wall is ruptured, the cytoplasm is dispersed, and this makes it impossible for the bacteria to reactivate. This is a process known as cell lysing.
Due to the fact that the ozone does not need to penetrate the cell wall, activated oxygen kills pathogens 3,125 times faster than chlorine, and without any chemical toxic residue left behind by the bacteria.
Virus
Ozone destroys viruses in a similar manner as bacteria. Ozone breaks apart lipid molecules at sides of multiple bond configurations, once the lipid envelope of the virus is fragmented and its DNA or RNA core they cannot survive.
With non-enveloped viruses, also called “naked viruses”, ozone reacts with the amino acids and capsid proteins, forming protein hydroxides and protein hydro peroxides.
Viruses have no defence against oxidative stress.
Fungi
While the fungus is more resistant to ozone than bacteria and viruses, ozone still gets the job done.
Fungus inhibited and destroyed by ozone include: Candida, Aspergillus, Histoplasma, Actinomycosis, and Cryptococcus. The presence of many disulfide bonds had been noted in these types of Fungi, making this a possible site for oxidative inactivation by ozone.
The Cleansing Power of Ozone: Activated Oxygen Oxidizes and Discomposes Matter. Ozone oxidises and/or ionises many forms of matter.
The great benefit of this process, considering practical applications, is ozone’s ability to alter, discompose and/or neutralise materials that are
toxic, without creating additional toxic compounds from the use of ozone (as is the case when using chemical compounds).
Ozone will oxidise metals (except gold, platinum, and iridium).